Medical device manufacturers looking to enter the Malaysian market must comply with regulatory requirements set by the Medical Device Authority (MDA). Understanding how to register a medical device in Malaysia is essential to ensure regulatory compliance and avoid delays in market entry.
Malaysia has established a structured regulatory framework under the Medical Device Act 2012 (Act 737), which governs the registration, licensing, and monitoring of medical devices. Before a medical device can be marketed or distributed in Malaysia, it must be registered with the MDA and comply with safety and quality standards.
In this guide, we explain the step-by-step process of medical device registration in Malaysia and what manufacturers need to prepare before submitting their application.
Key Takeaways
- Medical devices must be registered with Malaysia’s Medical Device Authority (MDA) before they can be marketed, imported, or distributed.
- Registration is governed by the Medical Device Act 2012 (Act 737) to ensure safety, quality, and regulatory compliance.
- Foreign manufacturers cannot apply directly and must appoint a Local Authorised Representative (LAR) in Malaysia.
- Devices are classified into Class A–D based on risk level, which determines documentation and assessment requirements.
- Class B, C, and D devices must undergo conformity assessment by a registered Conformity Assessment Body (CAB).
- Applications are submitted through the MeDC@St system with technical documentation and supporting evidence.
- Approval timelines vary depending on device classification and completeness of documentation.
- Working with experienced regulatory consultants can reduce delays and improve approval success.
What Is Medical Device Registration in Malaysia?
Medical device registration is the regulatory process required before a medical device can be sold, imported, or distributed in Malaysia. The registration ensures that the device meets safety, performance, and quality requirements established by the Medical Device Authority.
The regulatory framework aims to protect patient safety and ensure that only approved and compliant medical devices are available in the Malaysian healthcare market.
Manufacturers must submit their applications through the MeDC@St online system, which is the official portal used by the MDA for device registration and licensing.
Who Needs to Register Medical Devices?
Medical device registration applies to several parties involved in bringing medical devices into Malaysia.
These typically include:
- Medical device manufacturers
- Importers and distributors
- Local Authorized Representatives (LAR) representing foreign manufacturers
- Companies marketing medical devices in Malaysia
Foreign manufacturers cannot directly register their devices with the MDA. Instead, they must appoint a Local Authorised Representative (LAR) based in Malaysia to handle regulatory submissions and compliance matters.
Step-by-Step Process to Register a Medical Device in Malaysia
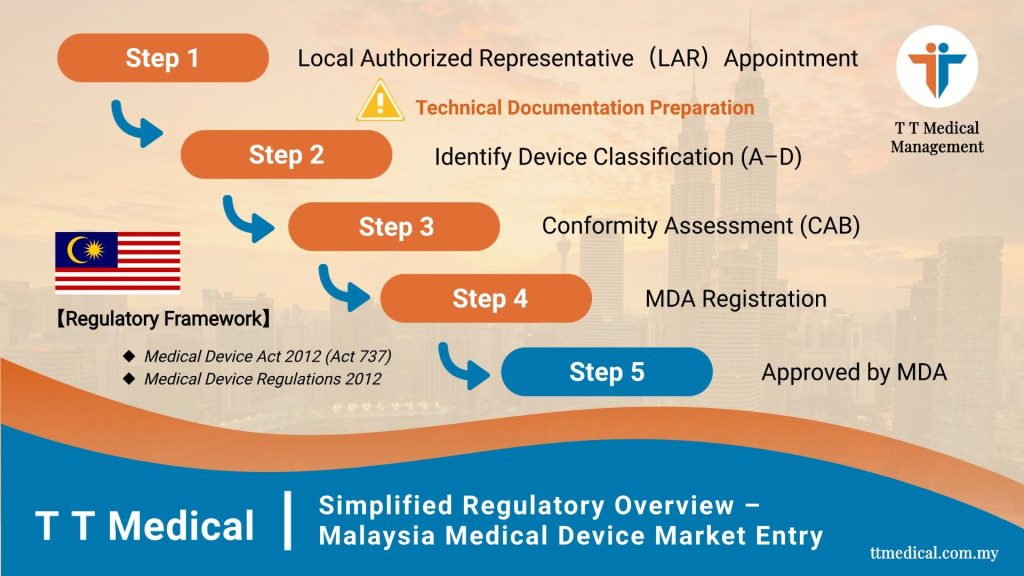
1. Determine the Medical Device Classification
Medical devices in Malaysia are classified based on risk level:
- Class A – Low risk devices
- Class B – Low to moderate risk
- Class C – Moderate to high risk
- Class D – High risk devices
The classification determines the level of regulatory scrutiny and documentation required for registration.
Higher-risk devices generally require a more comprehensive conformity assessment before approval.
2. Appoint a Local Authorised Representative (LAR)
Foreign manufacturers must appoint a Local Authorised Representative in Malaysia.
The LAR is responsible for:
- Submitting the medical device registration application
- Communicating with the Medical Device Authority
- Maintaining regulatory documentation
- Ensuring post-market compliance
The LAR acts as the official regulatory liaison between the manufacturer and the Malaysian authorities.
3. Prepare Required Documentation
To register a medical device in Malaysia, manufacturers must prepare technical documentation that demonstrates the safety and performance of the device.
Typical documents include:
- Device description and intended use
- Product specifications
- Risk analysis
- Clinical evaluation or performance data
- Quality management system certification (e.g., ISO 13485)
- Labeling and instructions for use
- Declaration of conformity
For higher-risk devices, additional technical documentation may be required.
4. Conduct Conformity Assessment
For Class B, C, and D devices, the application must undergo a conformity assessment conducted by a registered Conformity Assessment Body (CAB).
The CAB evaluates whether the device meets regulatory requirements before it can be submitted to the MDA.
This step helps ensure that medical devices meet international safety and quality standards.
5. Submit the Application via MeDC@St
Once all documentation is ready, the Local Authorised Representative submits the application through the MeDC@St system.
The submission typically includes:
- Device registration details
- Supporting technical documentation
- Conformity assessment reports (if required)
- Regulatory fees
The MDA will then review the application and request additional information if necessary.
6. Obtain MDA Approval
After the evaluation process is completed, the Medical Device Authority will issue the registration approval if all requirements are met.
Once approved, the medical device will be listed in the Medical Device Centralised Online Application System and can legally be marketed in Malaysia.
Manufacturers must also comply with post-market obligations such as vigilance reporting and regulatory updates.
Common Challenges in Medical Device Registration
Many manufacturers face challenges when navigating medical device registration in Malaysia. These challenges often include:
- Incorrect device classification
- Incomplete documentation
- Regulatory changes and updates
- Delays in conformity assessment
- Lack of local regulatory expertise
Working with experienced regulatory consultants can help ensure the process runs smoothly and reduce the risk of application rejection.
How Long Does Medical Device Registration Take?
The timeline for medical device registration depends on several factors, including the device classification and completeness of the documentation.
Generally:
- Class A devices may take a shorter approval period
- Class B–D devices require conformity assessment and longer review times
Proper preparation and regulatory expertise can significantly shorten the approval timeline.
How TT Medical Management Can Help
Navigating medical device registration requirements can be complex, especially for foreign manufacturers entering the Malaysian market.
TT Medical Management provides comprehensive regulatory support, including:
- Medical device registration services
- Local Authorised Representative services
- Regulatory consulting and compliance guidance
- Documentation preparation and submission
- Conformity assessment coordination
Our team has extensive experience assisting manufacturers in successfully registering their medical devices in Malaysia.
Learn more about our Medical Device Registration Services in Malaysia.
Conclusion
Understanding how to register a medical device in Malaysia is crucial for manufacturers seeking to enter the Malaysian healthcare market. The process involves device classification, regulatory documentation, conformity assessment, and submission through the MDA system.
By preparing the required documentation and working with experienced regulatory experts, manufacturers can streamline the registration process and ensure compliance with Malaysian regulations.


