If you’re planning to bring a healthcare product into Malaysia, chances are you’ve asked this at some point:
“Do I go through MDA or NPRA?”
It sounds like a small question, but getting it wrong can easily set you back a few months.
A lot of manufacturers assume the answer is obvious until they realise their product sits in a grey area. And by then, they’ve already started preparing the wrong submission.
Key Takeways
- MDA and NPRA regulate different types of healthcare products in Malaysia, each with its own requirements and process.
- The key difference between MDA & NPRA lies in whether the product works through a physical mechanism or a chemical effect.
- Misclassification is a common issue that can lead to delays, rework, and longer approval timelines.
- Not all health-related products fall under NPRA, as many are actually classified as medical devices under MDA.
- Getting clarity early helps save time, reduce costs, and avoid unnecessary back-and-forth.
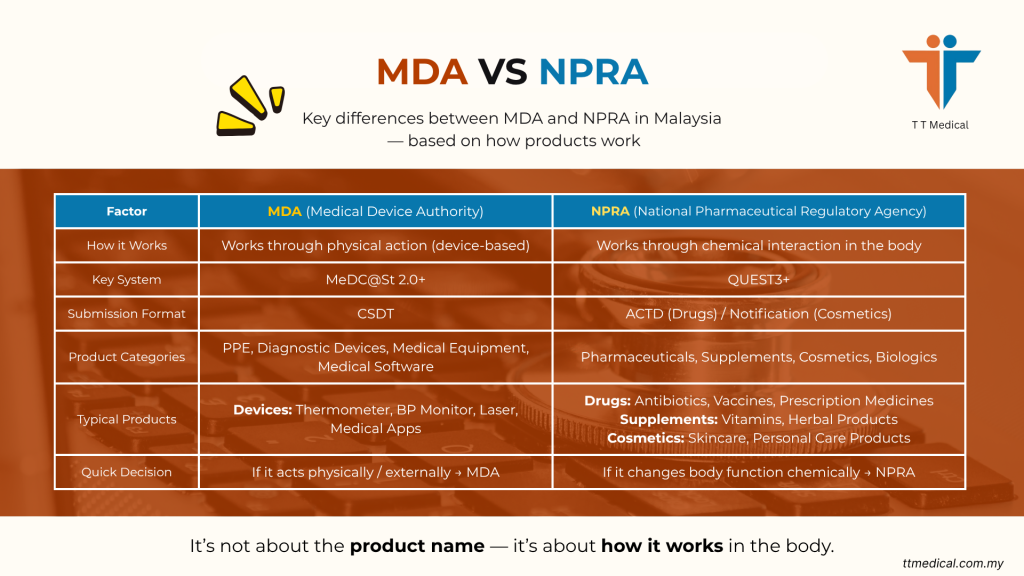
Overview: MDA vs NPRA in Malaysia
In Malaysia, healthcare products are mainly regulated by two bodies under the Ministry of Health:
To differentiate:
- MDA handles products that work in a physical way
NPRA handles products that work through chemical or biological effects
What is MDA (Medical Device Authority)?
The Medical Device Authority is responsible for regulating medical devices in Malaysia.
This generally includes products that operate through physical interaction, measurement, or mechanical function rather than chemical action.
What kind of products fall under MDA?
- PPE & Consumables: Surgical masks, gloves, and syringes.
- Diagnostic Tools: Thermometers and blood pressure monitors
- Aesthetic Tech: Lasers and HIFU devices (As of June 2026, these are under enhanced regulatory control).
- Software: Medical apps used for diagnosis or treatment.
The 2026 Process & Timelines
Applications are submitted through the MeDC@St 2.0+ system as part of the medical device registration process in Malaysia.
Devices are classified based on risk, ranging from Class A (low risk) to Class D (high risk).
If you’re unsure how your product is classified, it helps to understand the medical device classification framework in Malaysia before starting.
In terms of timelines:
- Lower-risk devices: Approved within a few months
- Higher-risk categories: Typically take longer, often up to a year
If your product has already been approved in markets like Singapore, certain reliance pathways may help shorten the process.
What is NPRA (National Pharmaceutical Regulatory Agency)?
The National Pharmaceutical Regulatory Agency is responsible for products that act inside the body through chemical or biological means
Typical products under NPRA
- Pharmaceuticals: Prescription drugs and OTC medicines.
- Supplements: Vitamins, minerals, and herbal remedies.
- Cosmetics & Skincare: Creams, makeup, and “functional” skincare.
- Biologics: Vaccines and blood products.
If your product changes how the body functions at a chemical level, NPRA is where you’ll be dealing with.
What does NPRA usually require?
Submissions go through the QUEST3+ system, and you’ll need the proper access set up before you can even start.
You’ll also have to put together a full ACTD dossier, covering things like safety, quality, and how the product actually works.
If the product sits in a grey area, you can request a classification check first to confirm which pathway to take.
Compared to MDA, NPRA usually looks more closely at the formulation and what effect the product has on the body.
Key Differences: MDA vs NPRA
|
Factor |
MDA (Medical Device Authority) |
NPRA (National Pharm. Regulatory) |
|
Primary Focus |
Physical/Mechanical performance |
Chemical/Biological effect |
|
Key System |
MeDC@St 2.0+ |
QUEST3+ |
|
Submission Format |
CSDT (Common Submission Dossier) |
ACTD (ASEAN Common Tech. Dossier) |
|
Main Concern |
Safety & Performance |
Safety, Efficacy & Quality |
|
Common Example |
Blood Pressure Monitor |
Blood Pressure Medication |
Where Most Manufacturers Get It Wrong
A lot of delays don’t come from the process itself, they happen when a product doesn’t clearly fit into one category.
- Medicated Patches: If the patch is mainly used to deliver a drug, it’s usually treated under NPRA.
- Dermal Fillers: Often seen as medical devices, but once ingredients like lidocaine are involved, they may be treated as combination products instead.
- Eye Drops: Simple lubricating drops are typically under MDA, but once you add antibiotics or steroids, they fall under NPRA.
This is where a lot of confusion starts, because the classification isn’t always obvious at first glance.
In these cases, it’s usually better to get a proper product classification assessment before submission to avoid delays later.
For these kinds of products, there’s also the Medical Device–Drug–Cosmetic Interphase Committee (MDDCI). If needed, your product may be reviewed there to confirm the correct category. It does add an extra step, but it helps avoid bigger issues later if the product is classified wrongly.
Why This Matters More Than You Think
Choosing the wrong route doesn’t just slow you down — it can affect your entire market entry plan.
We’re talking about:
- Delays of several months
- Missed distributor timelines
- Extra costs from re-submission
And if you’re coordinating multiple markets, one delay can throw everything off.
How TT Medical Can Help
If you’re not 100% sure where your product sits, you’re not alone this is exactly where most manufacturers need support.
TT Medical helps by:
- Clarifying whether your product falls under MDA or NPRA
- Preparing the right documentation from the start
- Managing submissions end-to-end
- Acting as your local representative in Malaysia
The goal is simple: get it right the first time, so you don’t lose months fixing avoidable mistakes.
FAQ
Approval timelines vary depending on product type and complexity. Medical devices can take a few months to up to a year, while NPRA timelines depend on the level of evaluation required.
The main difference is that Medical Device Authority (MDA) regulates medical devices, while National Pharmaceutical Regulatory Agency (NPRA) regulates pharmaceuticals, supplements, and cosmetics. The distinction depends on whether the product works physically or chemically.
It depends on the product’s primary mode of action. If it works through a physical or mechanical function, it typically falls under MDA, while products that act through chemical or biological effects are regulated by NPRA.
In most cases, a product is regulated by one authority. However, some hybrid products may fall into grey areas and require careful classification or further assessment before submission.
Switching between authorities is not straightforward and may require restarting parts of the process, which can significantly delay your approval timeline.


