If you’re planning to register a medical device in Malaysia, one of the first things you’ll need to figure out is your device classification.
It might sound like a small step, but it actually shapes everything from the documents you prepare to how long approval takes with the Medical Device Authority (MDA).
We’ve seen many cases where businesses get delayed simply because of incorrect classification. So in this guide, we’ll walk you through each class with clear, practical examples.
Key Takeways
- Medical devices in Malaysia are classified into Class A, B, C, and D based on risk level.
- The higher the class, the stricter the regulatory requirements and documentation.
- Classification is determined by intended use, duration, and level of invasiveness, not just the product itself.
- Misclassification is a common issue that can lead to delays, rework, and longer approval timelines.
- Getting the classification right early helps ensure a smoother registration process with the Medical Device Authority (MDA).
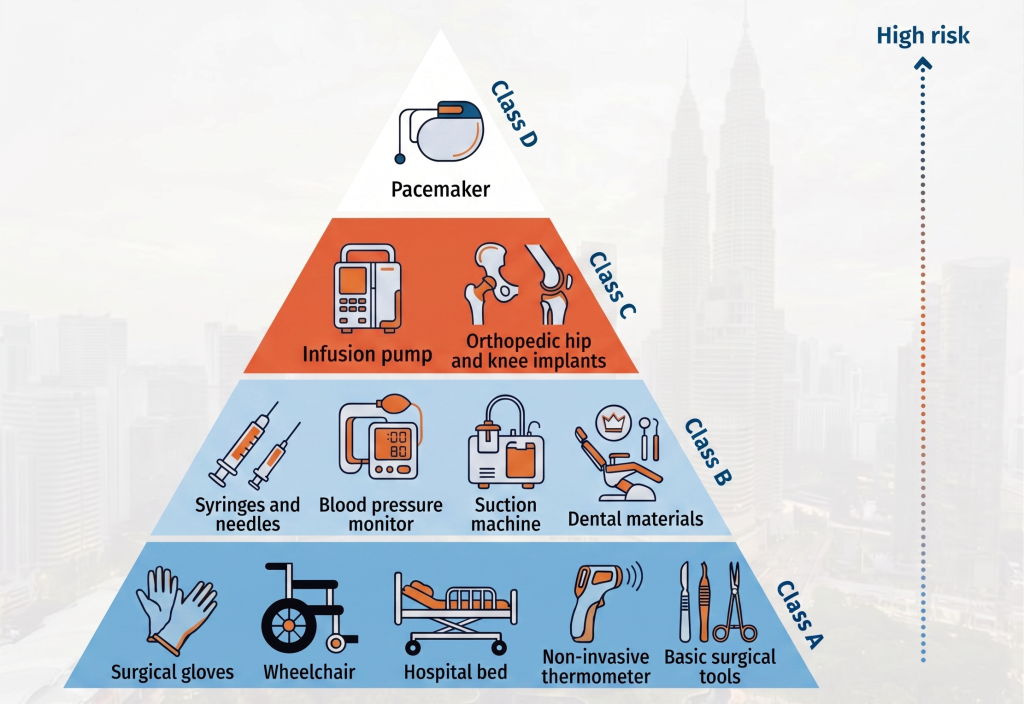
How Medical Devices Are Classified in Malaysia
In Malaysia, medical devices are grouped based on risk level:
- Class A – Low risk
- Class B – Low to moderate risk
- Class C – Moderate to high risk
- Class D – High risk
The classification depends on things like:
- What the device is used for
- How long it’s used on the body
- Whether it’s invasive
- The potential risk if something goes wrong
In simple terms — higher risk means stricter requirements.
Why Examples Matter in Device Classification
Understanding the rules is one thing. Applying them is another.
Two devices may look almost identical, but:
- one could be Class A
- the other could be Class B or even higher
The difference often comes down to:
- intended use
- duration of contact
- level of risk if the device fails
That’s why looking at real examples helps make classification clearer.
Typical Class A Devices You’ll See in Practice
Class A devices are generally straightforward and low risk. Most of them don’t interact deeply with the body.
Common examples
- Surgical gloves
- Wheelchairs
- Hospital beds
- Non-invasive thermometers
- Basic surgical tools
How to think about it
These devices are usually:
- External or non-invasive
- Used for general support rather than treatment
- Unlikely to cause harm if they malfunction
That said, “low risk” doesn’t mean “no rules”, they still need to meet regulatory requirements before entering the market.
Where Class B Devices Usually Apply
Class B devices step up slightly in terms of risk. They may interact with the body in a more direct way, but usually not for long periods.
Common examples
- Syringes and needles
- Blood pressure monitors
- Suction machines
- Dental materials
How to think about it
At this level:
- May enter the body briefly or assist in procedures
- Carry moderate risk if used incorrectly
- Require more structured documentation
This is where many companies start to feel the difference — the process becomes less “basic” and more structured.
When Devices Move Into Class C
Class C devices are used in more serious medical situations — often for treatment or ongoing monitoring.
Common examples
- Ventilators
- Infusion pumps
- Orthopedic implants
- Dialysis machines
Common examples
These devices:
- May remain in the body longer or support essential functions
- Have higher impact if performance fails
- Require formal conformity assessment
At this stage, having a clear regulatory plan can save you a lot of time.
High-Risk Devices Under Class D
Class D devices are the highest risk category. These are typically devices that support or sustain life.
Common examples
- Pacemakers
- Heart valves
- Implantable defibrillators
- High-risk diagnostic test kits
How to think about it
- Are critical to patient survival
- Require extensive evidence and evaluation
- Go through the strictest regulatory process
For these devices, there’s very little room for error during submission.
Common Grey Areas in Classification
Some devices don’t fall neatly into one category.
Examples include:
- Devices with both diagnostic and therapeutic functions
- Products with multiple intended uses
- Devices that vary based on duration of use
In these cases, classification depends heavily on how the product is positioned and described in your submission.
Common Mistakes Companies Make
From our experience at TT Medical, some of the most frequent issues include:
- Assuming similar products share the same class
- Overlooking intended use in classification
- Choosing a lower class to simplify the process
- Submitting without validating classification
These mistakes often result in:
- Reclassification by the authority
- Additional documentation requests
- Delays in approval timeline
How TT Medical Can Help
At TT Medical, we work closely with clients to make sure this step is done correctly from the start.
We support you with:
- Reviewing device specifications and intended use
- Advising on the most accurate classification approach
- Preparing documentation aligned with MDA expectations
- Supporting the full registration process
Whether you’re new to the Malaysian market or expanding your product line, we help make the process clearer and more manageable.
Final Thoughts
Device classification might seem like just another requirement, but it plays a big role in how smooth your registration process will be.
Getting it right early can save you time, cost, and unnecessary back-and-forth later on.
If you’re unsure where your device falls, it’s always better to check before moving forward.
FAQ
It depends on factors such as intended use, how long the device is used, and whether it is invasive. Devices with higher risk to patients are placed in higher classes.
No. While Class A devices have a simpler process, they still need to comply with regulatory requirements before being marketed in Malaysia.
Incorrect classification can lead to application delays, additional documentation requests, or even rejection. It may also increase your overall cost and timeline.
Yes. Similar products can be classified differently depending on their intended use. This is why classification should not be based on product type alone.
Generally, yes. Class C and Class D devices require more detailed evaluation, including technical documentation and conformity assessment, which can extend the approval timeline.
Yes. Clarifying your device classification early helps avoid unnecessary delays and ensures you prepare the correct documentation from the start.