If you're bringing a diagnostic test, laboratory reagent, or testing kit into Malaysia, you're dealing with a specific regulatory category that many manufacturers underestimate: In Vitro Diagnostics, or IVDs.
IVDs are regulated differently from standard medical devices in Malaysia. They have their own classification rules, their own documentation requirements, and their own risk logic — all governed by the Medical Device Authority (MDA) under the Medical Device Act 2012 (Act 737).
This guide walks through everything you need to know about IVD classification in Malaysia — what IVDs are, how they're classified, what each class requires, and the common mistakes that cause delays. If you're planning to register a medical device in Malaysia, understanding where your IVD fits is the most important first step.
Key Takeaways
- IVDs are medical devices used to examine specimens derived from the human body — they are not medicines.
- Malaysia classifies IVDs into four classes (A to D) based on the public health risk of an incorrect result.
- Class D IVDs carry the highest risk and require the most stringent documentation and review.
- IVD classification rules are separate from general medical device classification rules.
- All IVDs must be registered with the MDA before they can be imported, sold, or distributed in Malaysia.
- Foreign manufacturers must appoint a Local Authorised Representative (LAR) to handle the registration process.
- Misclassification is a common cause of submission delays and rework.
What Is an In Vitro Diagnostic (IVD) Device?
An In Vitro Diagnostic device is any medical device used to perform examinations on specimens derived from the human body — such as blood, urine, tissue, or other samples — with the purpose of providing information for diagnosis, monitoring, screening, or treatment decisions.
The term "in vitro" literally means "in glass" — referring to tests carried out outside the body, in a laboratory or clinical setting. IVDs do not directly interact with the patient's body during the test; they interact with biological specimens instead.
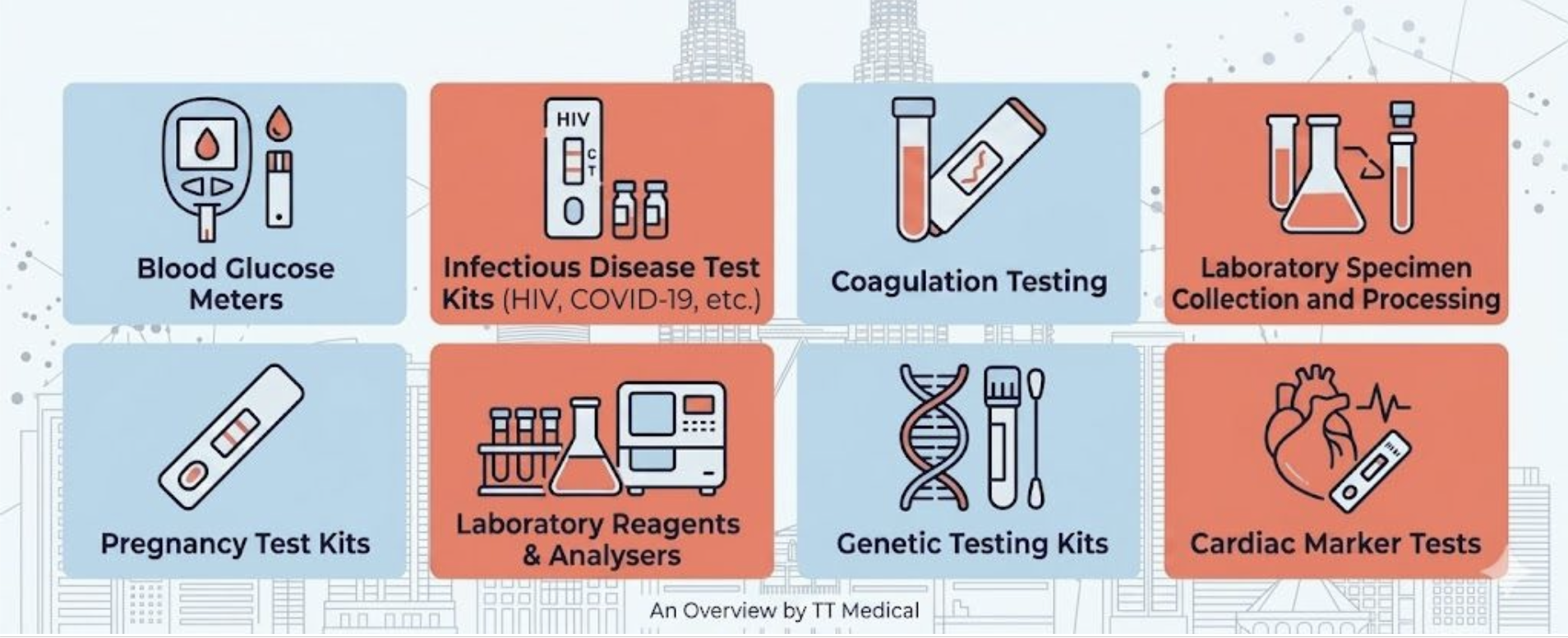
IVDs are not medicines or drugs. They are medical devices, which means they are regulated by the MDA under Act 737 — not by the NPRA. This distinction is important and is frequently a source of confusion during market entry.
Why It MattersWhy IVD Classification Works Differently
General medical devices are classified based on how they interact with the body — whether they're invasive, how long they're used, and whether they're active or non-active. IVDs, however, are classified based on a different set of principles entirely.
Because IVDs never directly enter or interact with the patient's body, the standard invasiveness-based rules don't apply. Instead, IVD classification in Malaysia is based primarily on the potential consequences of an incorrect test result — specifically the public health impact and clinical risk of a wrong diagnosis or missed detection.
The core question in IVD classification is: what happens if this test gives a false result? Tests where a wrong answer could lead to serious patient harm or widespread public health consequences are placed in higher risk classes.
A test kit that screens for the same condition can fall into different classes depending on whether it is designed for professional laboratory use, point-of-care use, or self-testing by the patient at home.
Whether the test is performed in a centralised laboratory, at the bedside, or by a patient at home affects the risk level. Self-tests and point-of-care tests may carry different classification requirements from the same test performed in a lab.
Tests used to screen blood donations or detect diseases with major public health implications are treated as higher risk, regardless of how technically straightforward the test itself may appear.
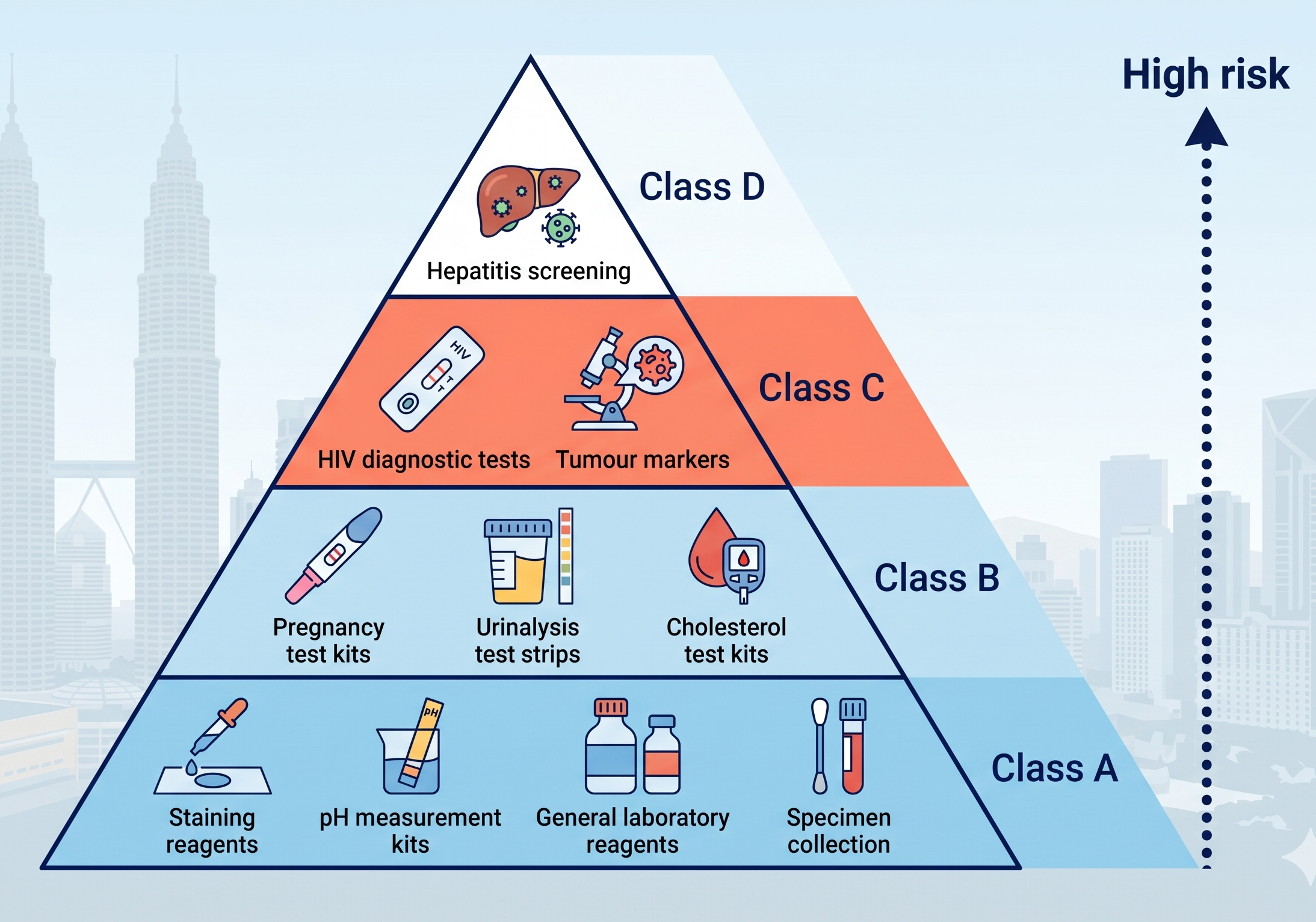
IVD Classification in Malaysia: Class A to D
Malaysia classifies IVDs into four classes under the same risk-based framework used for general medical devices, but applies IVD-specific classification rules. The higher the class, the greater the regulatory scrutiny and documentation required.
- General laboratory reagents
- Specimen collection and transport containers
- Culture media (general purpose)
- Staining reagents
- pH measurement kits
Class A IVDs pose minimal risk even if a result is incorrect. They are typically general-purpose laboratory tools not intended for specific disease diagnosis. They follow a simpler registration pathway but still require MDA registration before they can be marketed in Malaysia.
- Blood glucose monitoring systems
- Pregnancy test kits
- Urinalysis test strips
- Cholesterol test kits
- Ovulation prediction tests
Class B IVDs are used for diagnosing or monitoring specific conditions, but an incorrect result is unlikely to cause serious widespread harm. More structured technical documentation is required compared to Class A. Many self-test products commonly available in pharmacies fall into this category.
- HIV diagnostic tests
- Hepatitis B and C tests
- Tumour markers
- Antimicrobial susceptibility tests
- HbA1c tests for diabetes monitoring
Class C IVDs are used to diagnose serious infectious diseases or conditions where a missed or incorrect result could significantly harm the patient or contribute to disease spread. Conformity assessment by a registered CAB is required, along with comprehensive clinical performance data.
- Blood group typing reagents (ABO, Rhesus)
- Blood screening tests for transfusion safety
- HIV blood bank screening tests
- Hepatitis screening for blood donation
- Tests for Creutzfeldt-Jakob disease (CJD)
Class D IVDs pose the highest risk — a wrong result in blood screening or transfusion compatibility testing can directly cause patient death or widespread disease transmission. These require the most stringent evidence, the most comprehensive conformity assessment, and the most rigorous MDA review process.
IVD Classification vs General Medical Device Classification
| Factor | General Medical Devices | IVD Devices |
|---|---|---|
| Primary classification basis | Invasiveness, duration, active vs non-active | Risk of incorrect test result and public health impact |
| Patient contact | Direct contact with patient body | Indirect — tests biological specimens only |
| Classification rules | General device rules | IVD-specific rules under AMDD/MDA guidelines |
| Key documentation | Safety, performance, biocompatibility | Analytical performance, clinical performance, scientific validity |
| Self-test consideration | Not typically a classification factor | Self-test or lay-use status affects classification |
| Regulated by | MDA (Act 737) | MDA (Act 737) — same authority, separate rules |
What IVD Registration Requires in Malaysia
Registering an IVD in Malaysia follows the same overall framework as general medical device registration — but with IVD-specific documentation requirements. The key difference is in the technical evidence required to demonstrate the test performs as claimed.
Determine which class your IVD falls into based on intended use, specimen type, testing environment, and the public health risk of an incorrect result. This is the most critical step — getting it wrong affects everything that follows.
Foreign manufacturers must appoint an LAR in Malaysia. The LAR handles all MDA submissions, communications, and post-market compliance on your behalf.
IVD submissions require specific performance evidence in addition to standard technical documentation:
- Analytical performance data (sensitivity, specificity, precision)
- Clinical performance data and validation studies
- Scientific validity of the testing principle
- Risk management file (ISO 14971)
- Quality management system certification (ISO 13485)
- Labelling, instructions for use, and intended use statement
Class B, C, and D IVDs must be assessed by a registered Conformity Assessment Body (CAB) before submission to the MDA. Class D IVDs require the most comprehensive conformity review, including full evaluation of clinical performance data and quality systems.
The LAR submits the completed application through the MDA's MeDC@St 2.0 online system. Once approved, the IVD is listed in the MDA register and can be legally imported and distributed in Malaysia.
Common Mistakes in IVD Classification and Registration
IVD submissions face some specific pitfalls that differ from standard medical device registration. These are the issues we most commonly encounter when supporting clients through the process.
IVD Grey Areas That Catch Companies Off Guard
A diagnostic test designed to identify patients who are eligible for a specific drug treatment may be considered a companion diagnostic — and may require coordination with the NPRA if the drug is also being registered. Classification and regulatory pathway can become complex depending on how the products are positioned together.
The same test technology can fall into different classes depending on whether it is designed for a centralised laboratory, a bedside clinical setting, or home self-use. The testing environment affects the classification — a test validated in a lab setting is not automatically validated for self-testing.
Digital pathology tools, AI-based image analysis, and software that interprets IVD results may themselves qualify as medical devices or Software as a Medical Device (SaMD) — requiring separate registration. The IVD and the software component may each need their own MDA submission.
Products labelled "for research use only" are often assumed to be exempt from medical device regulation. However, if an RUO reagent is being sold into clinical settings or used to inform patient care decisions, it may require IVD registration regardless of its labelling.
IVD Registration Support from TT Medical
IVD registration in Malaysia requires a precise understanding of both the technical performance evidence needed and the classification rules that apply. TT Medical Management provides specialist support for IVD manufacturers navigating the MDA registration process.
Final Thoughts
IVD classification in Malaysia is not the same as general medical device classification. The rules are different, the documentation requirements are different, and the risk logic is different. Understanding these differences before you begin is the most reliable way to avoid delays and ensure your submission is right the first time.
Whether your product is a simple laboratory reagent or a high-risk blood screening test, getting the classification confirmed early — before you've invested in the wrong documentation — will save significant time and cost. If you're unsure where your IVD fits, or if it sits in a grey area, speak to our consultancy team before moving forward.
FAQ